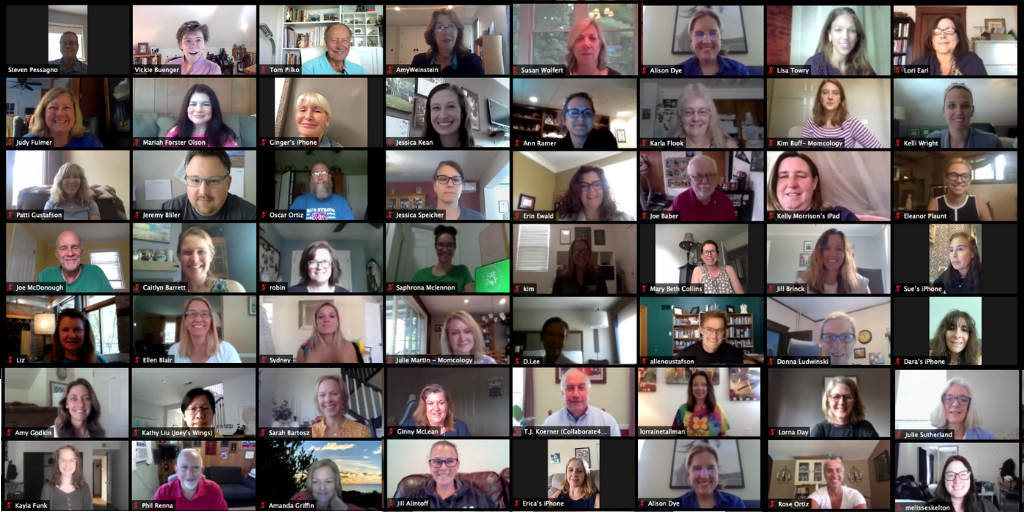
Over 100 CAC2 members and guests gathered for a special online event on Friday, July 17, 2020 to attend our 8th Annual Summit Speaker Program and Annual Members’ Business Meeting. Our volunteer Meetings Team designed a carefully curated two-hour version of our traditional Annual Summit accessible to all.
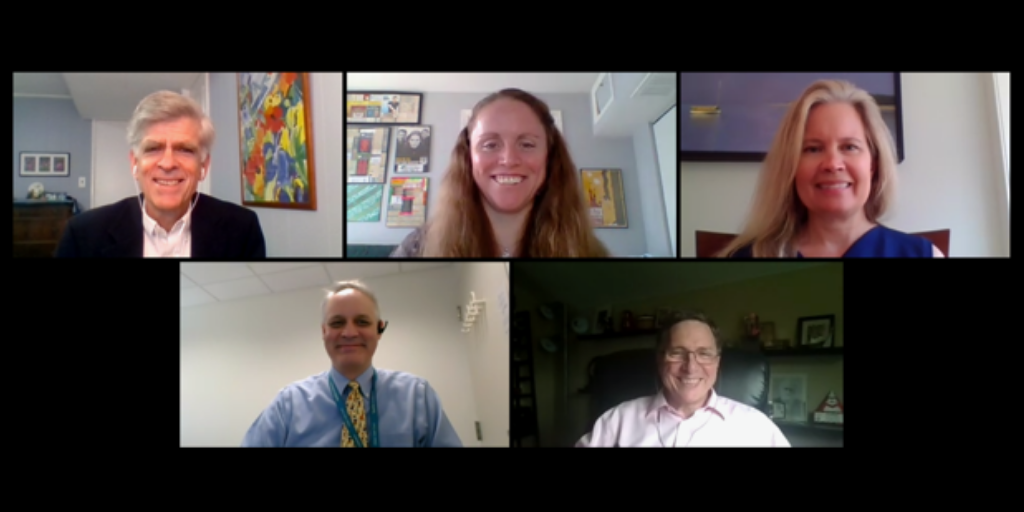
- Dr. Doug Hawkins, newly elected Chair of the Children’s Oncology Group, speaking on his vision for COG, with Q&A on how parent patient advocates fit into that vision
and
- The RACE Act on the Eve of Implementation Panel, a conversation moderated by Dr. Stacey Adam (FNIH) featuring Dr. Malcolm Smith (NCI), Dr. Martha Donoghue (FDA), and Dr. Mark Kieran (Bristol Myers Squibb) discussing the possibilities for pediatric oncology drug development presented by regulations going into effect in August.
We are pleased to share the video recording of our speaker program:
Dr. Hawkins talk elicited so much engagement that we couldn’t get to all the questions and comments. He graciously agreed to video another session so that everyone’s questions could be addressed.
We hope to gather in person June 24-26, 2021 at Children’s Hospital of Philadelphia. In the meantime, we were thrilled to see each other!
Categories
- Blog Posts (91)
- Explainer Post (11)
- Guest Blogs (15)
- Member Blogs (37)
- Survivorship Matters Blog (9)
- CAC2 News (25)
- Collaborative Achievements (13)
- Community News (233)
- In Memoriam (2)
- News Digest (186)
- Webinars (64)
Tags
#CancelChildhoodCancer (1)
Advocacy (55)
Annual Summit (7)
Appropriations (1)
Awareness (26)
AYA (2)
Bereavement (1)
Best Practices (14)
Biorepositories (2)
CAC2 News (9)
CAC2 Project Incubator (1)
CAC2 Projects (11)
Call to Action (25)
Call to Action Prevention (1)
Clinical Trials (11)
COG (2)
Collaboration (13)
Data (15)
Delayed Diagnosis (2)
Digest (12)
Drug Development (11)
Drug Development collaboration (1)
Drug Shortages (2)
Ethics (1)
Family Support (16)
Funding Research (7)
Genomics (6)
Global News (3)
Industry Perspective (5)
Member Access (12)
NCI (3)
Nutrition (2)
Patient-Reported Outcomes (1)
Post-Mortem Tissue Donation (1)
Prevention (4)
Psychosocial Support (6)
Rare and Ultra Rare Cancer (1)
Rare Cancers (1)
Regulations (3)
Research (36)
Research Data (2)
Research Tissue Donation (1)
Social Work (1)
Survivorship (28)
Venture Philanthropy (3)
Recent Posts
- CAC2 Childhood Cancer Community News Digest (July 15-21)
- CAC2 Collaborative Achievement–CAC2 Members Build Framework for Advancing Pediatric Cancer Research and Drug Development
- CAC2 Childhood Cancer Community News Digest (July 8-14)
- CAC2 Childhood Cancer Community News Digest (July 1-7)
- NCI Advocacy Update